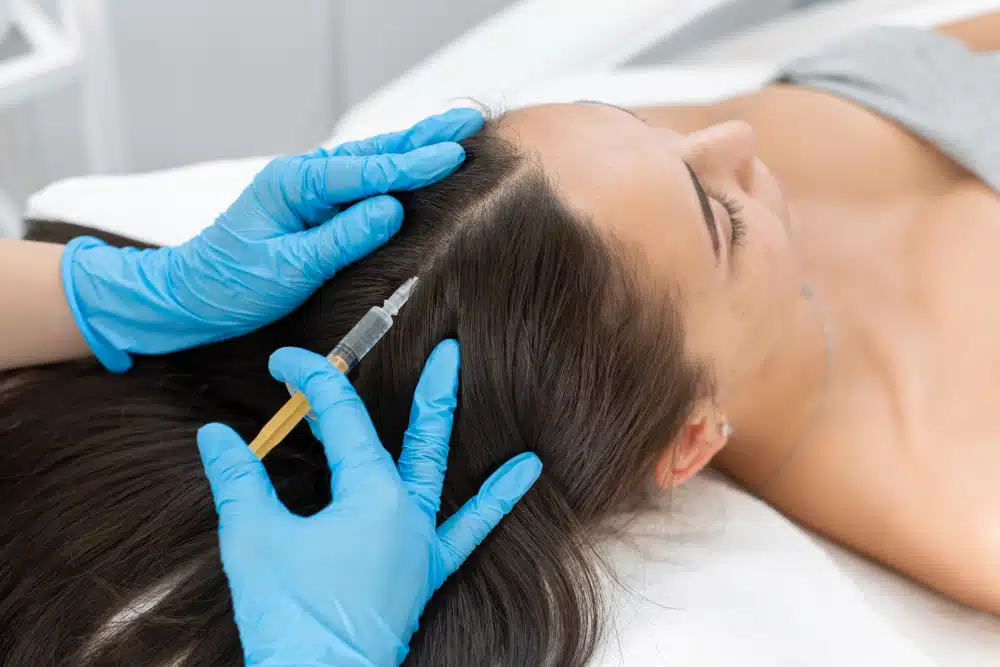
Dutasteride
mesotherapy
for hair loss.
A physician-administered intradermal injection that delivers a potent DHT blocker directly into the scalp, targeting androgenetic alopecia at its hormonal root without systemic exposure.

Targeting hair loss
at its hormonal root.
Androgenetic alopecia (AGA) is driven by dihydrotestosterone (DHT), a hormone that binds to androgen receptors in scalp follicles and causes them to miniaturize over time, producing progressively finer, shorter hairs until the follicle is no longer active. Dutasteride works by blocking the enzyme that converts testosterone into DHT, interrupting this miniaturization process.
Mesotherapy is the delivery method: a series of micro-injections that deposit dutasteride directly into the intradermal layer of the scalp, placing the active drug precisely where the follicles are affected. This targeted approach aims to build meaningful local drug concentration while avoiding the hormonal disruption that comes with taking dutasteride orally.
Unlike topical treatments that depend on skin penetration, and unlike oral tablets that affect the whole body, dutasteride mesotherapy offers a middle path: precise local delivery with physician oversight at every session.
See why dutasteride
mesotherapy is emerging
as a leading option.
This short video explains the science behind dutasteride mesotherapy for androgenetic alopecia, what the treatment involves, and the results patients are experiencing. It is the most efficient way to understand whether this approach fits your hair loss pattern before booking a consultation.
Dutasteride mesotherapy is an off-label treatment. During your consultation, Dr. Akbari will review your hair loss type, severity, and medical history to confirm whether this is an appropriate option for you and outline exactly what to expect.
How dutasteride
stops follicle
miniaturization.
Hair follicles contain androgen receptors that are sensitive to dihydrotestosterone (DHT). When DHT binds to these receptors, it shortens the anagen (growth) phase and progressively shrinks the follicle, producing thinner, shorter hairs with each cycle until the follicle stops producing visible hair entirely.
The enzyme 5-alpha-reductase converts testosterone into DHT. Finasteride blocks only the type II isoform of this enzyme. Dutasteride blocks both type I and type II, producing a more complete reduction in local DHT levels. This is why dutasteride is generally considered more potent than finasteride for androgenetic alopecia treatment.
When delivered via mesotherapy, the injected solution is deposited into the dermis of the scalp using fine 30-gauge needles spaced 0.5 to 1 cm apart. The drug builds local concentration in the tissue surrounding the follicles, acting on the enzyme pathway at the site where it matters, with a substantially lower dose than required for systemic oral administration.
Improved studies suggest that when sessions are frequent enough and the protocol is adequately maintained, measurable increases in hair density and shaft diameter can be achieved. The best outcomes are typically seen when mesotherapy is used as part of a broader hair restoration protocol tailored to the individual.
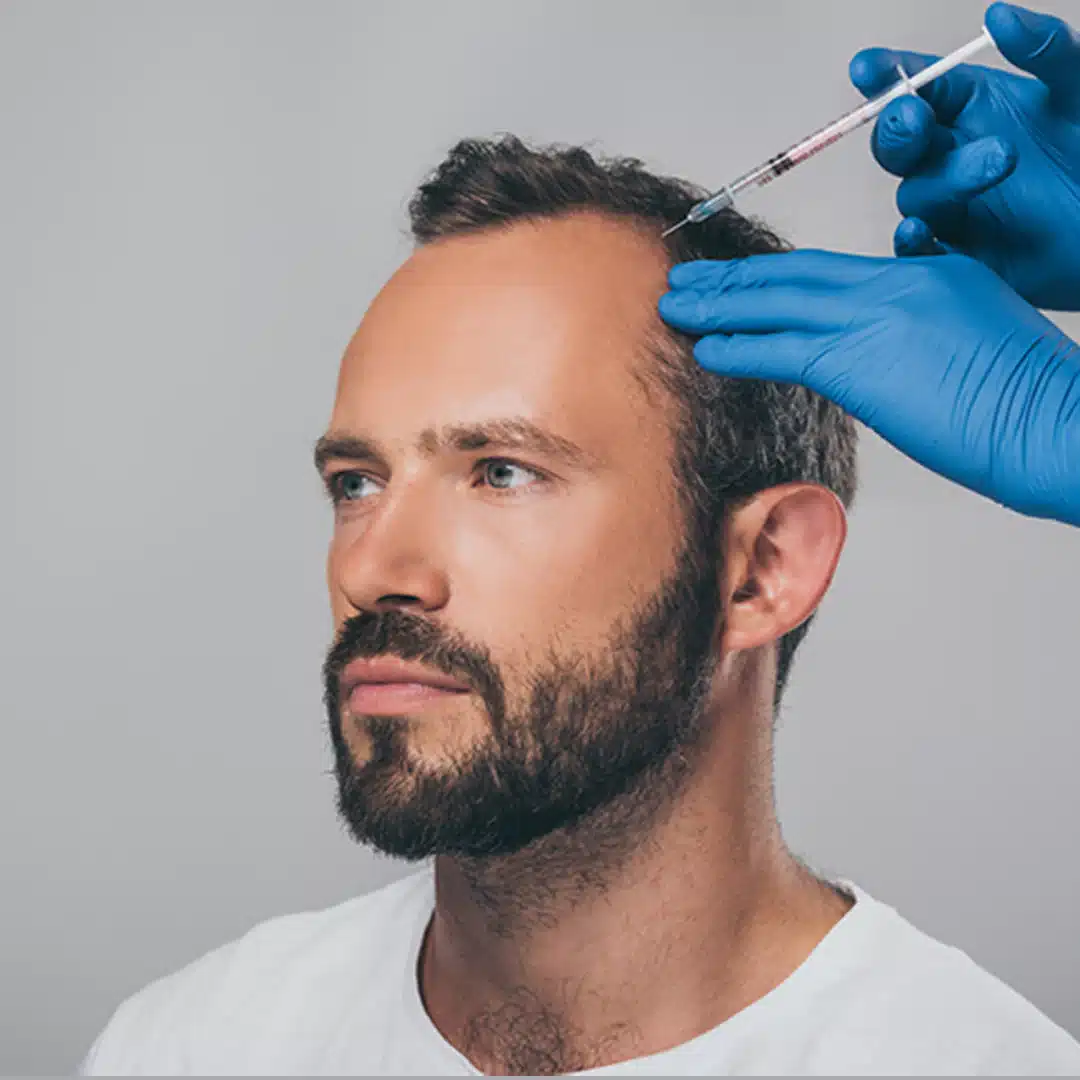
Dutasteride mesotherapy
vs. the alternatives.
Mesotherapy
What a dutasteride
mesotherapy course looks like.
-
01
Hair Loss Consultation
Dr. Akbari reviews your hair loss type, Norwood or Ludwig grading, medical history, and current treatments. Trichoscopy may be used to assess follicle density and miniaturization. Together you confirm whether dutasteride mesotherapy is the appropriate choice, and whether it will be used alone or as part of a broader protocol. -
02
Session 1 (Month 0)
A dilute dutasteride solution is injected intradermally across the affected scalp using a 30-gauge needle, with injection points spaced 0.5 to 1 cm apart. Each session takes approximately 20 to 30 minutes. No anaesthesia required for most patients, though topical numbing is available. -
03
Sessions 2 and 3 (Months 3 and 6)
Follow-up sessions build on the initial treatment. The most widely studied protocol spaces sessions every 3 months. Hair cycling means visible improvements develop gradually over several months, not immediately after each injection. -
04
Assessment at Month 9
Response is formally evaluated 3 months after the final session. Trichoscopy measurements of hair density and diameter are compared to baseline. This timeline reflects how hair cycles respond to DHT reduction: follicles need several months to begin producing thicker, longer hairs again. -
05
Maintenance and Ongoing Care
Based on your response, a maintenance protocol is discussed. Some patients continue quarterly sessions long-term. Others transition to or combine with complementary treatments such as PRP, topical minoxidil, or low-level laser therapy to sustain and build on results.
Ideal candidates for
dutasteride mesotherapy.
Who should not
proceed without
physician review.
A physician evaluation is required before any treatment. Dutasteride mesotherapy is not appropriate for all patients. The following groups require special consideration or are excluded:
What the clinical
literature shows.
Real patients.
Real regrowth.
"I had been on finasteride for two years with limited results and started worrying about side effects. Dr. Akbari suggested dutasteride mesotherapy as an alternative. Nine months in and the density at my crown is noticeably better. I appreciate that it is supervised and measured at every visit."
"As a woman I was told for years that there was nothing really effective I could take for hair thinning. Dutasteride mesotherapy was presented to me as a targeted option that bypasses a lot of the concerns with oral medications. I have completed three sessions and the texture and thickness of my existing hair has genuinely improved."
"The consultation was extremely thorough. Dr. Akbari explained the science, the off-label status, what results are realistic and what the evidence actually shows. I felt like I was making an informed decision, not just being sold something. The injections themselves are quick and easy."
Your questions,
answered.
Dutasteride mesotherapy is an intradermal injection treatment for androgenetic alopecia (male and female pattern hair loss). A dilute solution of dutasteride is injected into the scalp using fine 30-gauge needles spaced 0.5 to 1 cm apart, depositing the drug directly into the layer of tissue where the hair follicles reside.
Dutasteride is a dual 5-alpha-reductase inhibitor that blocks the conversion of testosterone into dihydrotestosterone (DHT). DHT is the hormone responsible for follicular miniaturization in AGA. By reducing local DHT levels in the scalp, the treatment aims to slow or halt miniaturization and allow existing follicles to produce thicker, longer hairs over time.
Finasteride inhibits only type II 5-alpha-reductase. Dutasteride inhibits both type I and type II. Since both isoforms are present in scalp follicles, dutasteride's dual inhibition produces more complete DHT suppression in the scalp tissue.
Clinical network meta-analyses have consistently ranked oral dutasteride ahead of finasteride for improving total hair density in men with AGA. The trade-off historically has been that dutasteride's much longer half-life (approximately 5 weeks vs. finasteride's 6 to 8 hours) means any side effects take longer to resolve after stopping the medication, which is one reason some patients and clinicians prefer the mesotherapy route.
The most widely studied protocol consists of 3 sessions spaced approximately 3 months apart, with response formally evaluated at 9 months from the first injection. Some earlier published protocols used more frequent initial sessions, spaced 1 to 4 weeks apart, before moving to quarterly maintenance.
Results develop gradually because hair cycling takes time to reflect changes in the follicular environment. Most patients begin noticing a change in hair texture and shedding rate before they see measurable density improvement. The 9-month assessment timeline aligns with the biology of how follicles respond to reduced DHT exposure.
A key advantage proposed for mesotherapy over oral dutasteride is reduced systemic absorption. In a 2017 prospective study using a quarterly session protocol, researchers detected no statistically significant differences in serum hormone levels before and after the treatment course, suggesting limited systemic absorption at the studied dose and frequency.
However, the scalp is highly vascular and some studies have proposed that absorption may be more significant with frequent sessions or higher doses. The safety profile in the largest real-world cohort to date (541 patients) was described as favorable. All patients at Monarch MD are evaluated before treatment and followed throughout their protocol.
Yes. Dutasteride mesotherapy has been studied in both men and women with AGA. A 2013 study specifically evaluated mesotherapy using a dutasteride-containing preparation for female pattern hair loss, reporting improvements in hair density and structure. Women who are not pregnant and not planning to become pregnant are generally considered potential candidates after physician evaluation.
Dutasteride carries a teratogenic risk and is strictly contraindicated in pregnancy. Any woman of childbearing potential should discuss this with Dr. Akbari before beginning treatment. Reliable contraception is required during treatment for this patient group.
Dutasteride is approved by Health Canada and the FDA for benign prostatic hyperplasia. It is not specifically approved by Health Canada or the FDA for androgenetic alopecia, meaning its use for hair loss is off-label. Dutasteride has been approved for AGA in South Korea, Japan, and certain other countries following clinical trials specifically for this indication.
Off-label prescribing is a legal and common medical practice in Canada when a physician determines that the evidence supports the treatment for a given patient. Dr. Akbari will review the clinical evidence and your individual case during consultation so that you can make a fully informed decision.
Yes. Dutasteride mesotherapy for androgenetic alopecia is available at Monarch MD Facial Plastic Surgery, located at 1553 Marine Drive, West Vancouver, BC. Treatment is physician-supervised and preceded by a comprehensive hair loss consultation. We serve patients from West Vancouver, North Vancouver, Burnaby, and greater Vancouver.
You can book your consultation online or call us directly. We recommend beginning with our comprehensive hair analysis so that dutasteride mesotherapy, if appropriate, is planned as part of a full hair restoration strategy rather than in isolation.
Start with a
hair analysis.
Before any treatment begins, we assess your hair loss type, severity, and medical history. Dutasteride mesotherapy may be one part of your protocol, or the cornerstone of it. Find out what is right for you.
This page is for educational purposes only and does not constitute medical advice. Dutasteride mesotherapy is an off-label treatment for androgenetic alopecia and requires a physician consultation and informed consent. Dutasteride is contraindicated in pregnancy and in women who may become pregnant. Results vary by individual. All treatments at Monarch MD are preceded by a medical evaluation. Content reviewed by Dr. Eli Akbari, MD, FRCSC, Facial Plastic Surgeon, Monarch MD Facial Plastic Surgery, West Vancouver, BC. Learn about Dr. Akbari · Comprehensive Hair Analysis